🔄 AKI-AKD-CKD Continuum
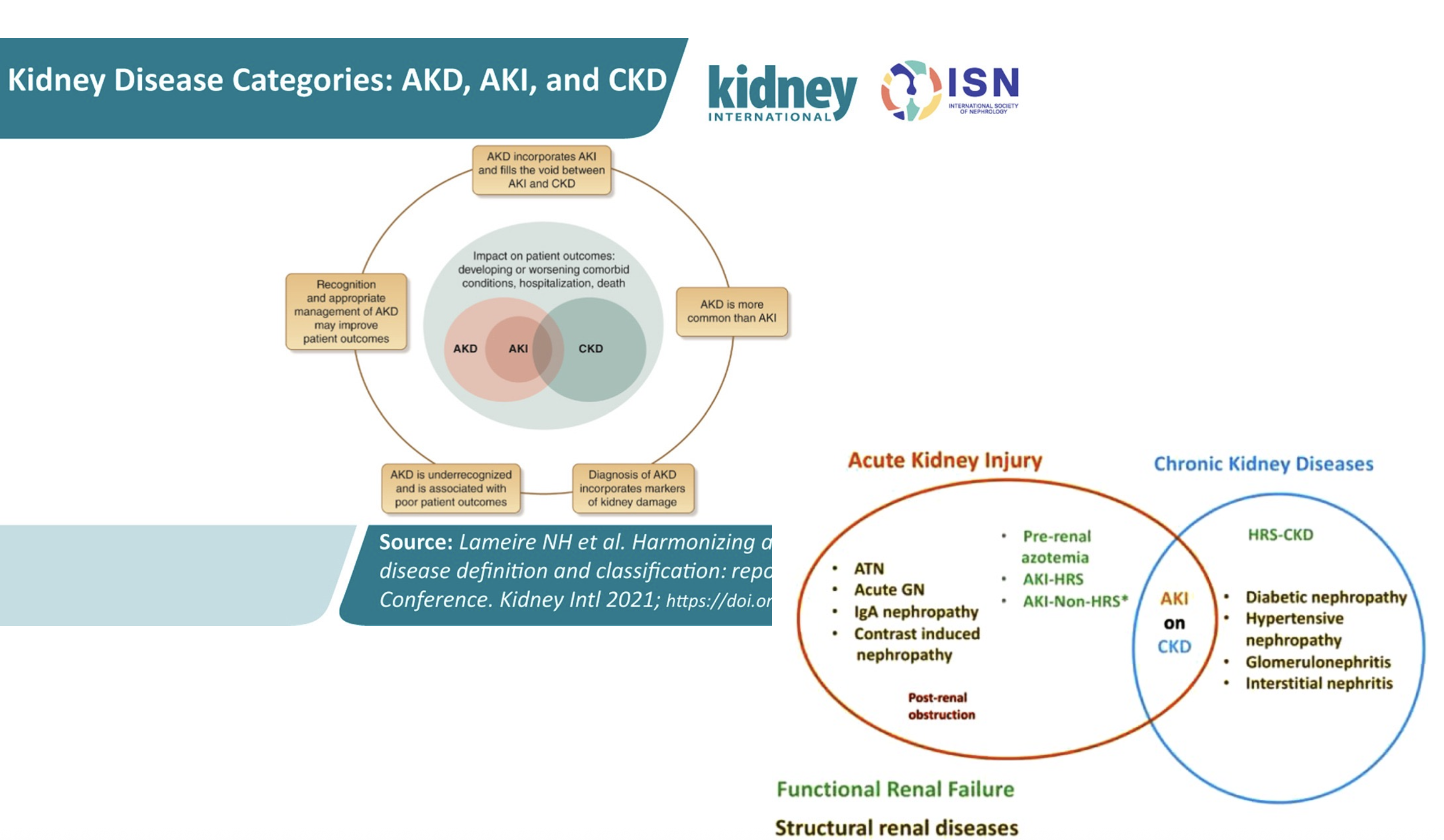
Modern understanding recognizes kidney disease as a continuum rather than discrete categories, with acute kidney disease (AKD) serving as the bridge between acute injury and chronic disease.
📋 Acute Kidney Disease (AKD) — Formal Definition (KDIGO 2026)
AKD is defined as any of the following persisting for ≤3 months:
- AKI (functional or structural criteria)
- GFR <60 mL/min/1.73m²
- GFR decrease ≥35 mL/min from baseline
- SCr increase >50%
- Markers of kidney damage (albuminuria, hematuria, leukocyturia)
🚨 Emergency AKI Recognition Protocol
Time-sensitive evaluation for rapid clinical decision-making
Vital signs, volume status, bladder scan, 12-lead ECG
- Check for hyperkalemia (peaked T waves)
- Assess hemodynamic stability
- Rule out obstruction first
BMP, CBC, urinalysis with microscopy, ECG
- Repeat K+ if >5.5 mEq/L
- Rule out hemolysis
- Check magnesium and phosphorus
If bladder scan >150mL or unexplained AKI
- May resolve AKI completely
- Check post-void residual
- Monitor urine output closely
Fresh specimen within 2 hours of collection
- Differentiate prerenal vs intrarenal vs postrenal
- Look for casts, crystals, cells
📊 KDIGO AKI Staging (2026 Three-Axis System)
Staging now incorporates three independent axes: Serum Creatinine (C), Urine Output (U), and Damage Biomarker (B)
| Stage | Serum Creatinine (C) | Urine Output (U) | Damage Biomarker (B) | Clinical Implications |
|---|---|---|---|---|
| C0/U0/B1 | No SCr or UOP change | No UOP change | B1 = Positive | Subclinical AKI — biomarker-only detection, intervene early |
| C1/U1 | ≥0.3 mg/dL or 1.5-1.9× baseline | <0.5 mL/kg/h (ideal BW) for 6-12h | B0 or B1 | Early recognition, identify etiology |
| C2/U2 | 2.0-2.9× baseline | <0.5 mL/kg/h for >12h | B0 or B1 | Increased monitoring, nephrology consult |
| C3/U3 | ≥3.0× baseline, OR ≥4.0 mg/dL, OR RRT | <0.3 mL/kg/h for >24h OR anuria >12h | B0 or B1 | Consider RRT, ICU monitoring |
⏲ Transient vs. Persistent AKI (KDIGO 2026)
| Category | Definition |
|---|---|
| Transient AKI | ≤48h increase in SCr or cystatin C, or reduced UOP |
| Persistent AKI | >48h and up to 7 days |
🔁 Recurrent AKI (KDIGO 2026)
Recurrent AKI is defined as a new episode after partial or complete resolution of a previous episode. Recurrent AKI confers additional risk for CKD progression and cardiovascular mortality, and should prompt evaluation for modifiable risk factors and closer long-term follow-up.
📐 Baseline SCr Selection — Hierarchical Approach (KDIGO 2026 Figure 1)
🧐 Critical Concept: Why eGFR is Invalid in AKI
Fundamental Principle: eGFR calculations are only valid when serum creatinine is at steady state
🔴 AKI: Rising Creatinine = Invalid eGFR
- Creatinine Status: Rapidly rising (non-steady state)
- eGFR Validity: ❌ INVALID - significantly overestimates true GFR
- Clinical Reality: True GFR may be near zero while eGFR still appears "normal"
- Time to Steady State: 3-5 days after GFR change
- Clinical Use: Monitor creatinine trends, not absolute eGFR values
🟢 CKD: Stable Creatinine = Valid eGFR
- Creatinine Status: Stable over months (steady state)
- eGFR Validity: ✅ VALID - accurately reflects true GFR
- Clinical Reality: eGFR closely approximates measured GFR
- Time Course: Gradual changes over months to years
- Clinical Use: Staging, monitoring, medication dosing
🧬 The Total Nephrectomy Teaching Example
Scenario: Patient undergoes total nephrectomy (both kidneys removed)
⚡ Immediate Reality (Time = 0)
- True GFR: 0 mL/min (no kidneys!)
- Serum Creatinine: Still normal (1.0 mg/dL)
- Calculated eGFR: ~90 mL/min (completely wrong!)
- Clinical Status: Patient is anuric, requires emergency dialysis
🕰️ Day 1 Post-Op
- True GFR: Still 0 mL/min (unchanged)
- Serum Creatinine: 2.0 mg/dL (rising)
- Calculated eGFR: ~38 mL/min (still way off!)
- Clinical Status: Same - still requires dialysis
📈 Day 2 Post-Op
- True GFR: Still 0 mL/min (unchanged)
- Serum Creatinine: 3.5 mg/dL (continuing to rise)
- Calculated eGFR: ~20 mL/min (getting closer but still wrong)
- Clinical Status: Same - still requires dialysis
📊 Day 3 Post-Op
- True GFR: Still 0 mL/min (unchanged)
- Serum Creatinine: 5.0 mg/dL (continuing upward)
- Calculated eGFR: ~13 mL/min (improving but still overestimating)
- Clinical Status: Same - still requires dialysis
🎯 Steady State (Days 4-5)
- True GFR: Still 0 mL/min (unchanged)
- Serum Creatinine: Plateaus (8-12 mg/dL)
- Calculated eGFR: ~5-7 mL/min (finally approaching reality)
- Clinical Status: Same - chronic dialysis needed
💡 Key Teaching Points
- GFR changed instantly (0 mL/min immediately after surgery)
- Creatinine rises gradually over 3-5 days to reach new steady state
- eGFR "catches up" slowly and significantly lags behind true GFR changes
- Clinical decisions must be based on the clinical scenario, not misleading eGFR values
- In AKI, trend and absolute creatinine values matter more than calculated eGFR
🎯 Clinical Applications in AKI Management
❌ Don't Rely On:
- eGFR values during rising creatinine
- "Normal" eGFR in early AKI
- eGFR for medication dosing in AKI
- eGFR for dialysis timing decisions
✅ Instead Focus On:
- Absolute creatinine values and trends
- KDIGO staging criteria
- Clinical signs: urine output, volume status
- Time course and rate of creatinine rise
👥 Patient Education:
- Explain why eGFR seems "better" than reality
- Focus on kidney function recovery, not numbers
- Emphasize clinical improvement markers
- Avoid false reassurance from "improving" eGFR
⚠️ Common Clinical Errors to Avoid
- "The eGFR is still 45, so the kidneys are working okay" - Wrong! In AKI, this could represent severe kidney injury
- Using eGFR for medication dosing in AKI - Can lead to significant overdosing
- Reassuring patients based on "stable" eGFR - While creatinine is still rising
- Delaying nephrology consultation - Because eGFR "doesn't look that bad"
- Comparing AKI eGFR to CKD staging - Fundamentally different clinical contexts
Do NOT Use the eGFR from the BMP for Drug Dosing in AKI
The eGFR printed on your BMP is a steady-state equation (CKD-EPI). It is only valid when creatinine is stable. When creatinine is actively rising, that number OVERESTIMATES true GFR — potentially by a massive margin. A patient whose Cr jumped from 1.0 to 2.0 in 24 hours may have a true GFR near zero, but the BMP prints “eGFR 38” — this is WRONG.
Overdosing of renally cleared drugs because the “eGFR looks okay”
“The eGFR is 45, kidneys are fine” — while GFR may actually be near zero
Consult deferred because the eGFR “doesn't look that bad”
✅ Kinetic eGFR (KeGFR) — A Better Estimate, But Not a Magic Number
KDIGO 2026 Rec 2.3.2 (2B) KDIGO 2026 formally recommends calculating kinetic eGFR (KeGFR) in hospitalized adults when a non-steady-state GFR estimate is needed.
KeGFR accounts for the rate of creatinine change, providing a more accurate real-time GFR estimate during AKI:
- Uses the rate of SCr rise (ΔCr/Δtime) to estimate true GFR
- More accurate than steady-state eGFR when creatinine is rapidly changing
- Useful for drug dosing decisions in AKI
- Valuable for assessing AKI recovery trajectory
⚠️ KeGFR Caveats — Evidence Is Limited
- The evidence base is small — limited validation studies, no large RCTs confirming KeGFR-guided decisions improve outcomes
- The formula assumes stable creatinine production, which is NOT true in:
- Sepsis — altered muscle metabolism
- Muscle wasting / critical illness — reduced Cr generation
- Rhabdomyolysis — massive Cr release from muscle
- Large volume resuscitation — dilutional effects
- KDIGO grades this 2B — a weak recommendation with moderate certainty
🎯 The More Important Clinical Lesson
You do not need to calculate KeGFR at the bedside. What you DO need to do is IGNORE the eGFR on the BMP when creatinine is changing.
The TREND IS the GFR estimate. A rising creatinine = GFR is lower than any equation tells you. A falling creatinine = GFR is recovering. Use clinical judgment and creatinine trends for drug dosing — not the number the lab prints.
❌ Do NOT
Dose vancomycin, aminoglycosides, or DOACs based on the eGFR printed on a BMP when Cr is rising
✅ DO
Use creatinine trajectory + clinical context. When in doubt, dose for a lower GFR than the equation suggests.
🔍 Systematic Diagnostic Approach
🔬 Urine Microscopy: The Key Differentiator
Fresh urine microscopy within 2 hours provides critical diagnostic information
💧 Prerenal AKI
🔥 Intrarenal AKI
| ATN | Muddy brown casts, RTEs |
| GN | RBC casts, dysmorphic RBCs |
| AIN | WBC casts, sterile pyuria |
🚰 Postrenal AKI
🔊 Kidney Doppler Ultrasound & RRI
Measurement of renal resistive index (RRI) may aid in risk stratification for AKI progression and severity, particularly in critically ill populations.
🛡️ Kidney Functional Reserve
Assessment of kidney functional reserve before a potential nephrotoxic exposure may help identify patients at increased risk of developing AKI.
⚠️ FeNa and FeUrea — Know Their Limits
These are commonly over-relied-upon tests. Understand exactly what they can and cannot tell you.
The ONLY Clinical Question FeNa Reliably Answers
In the oliguric patient: is this prerenal (hemodynamic) AKI or ATN?
That is the narrow clinical scenario where FeNa has reasonable diagnostic performance. Outside of this context, FeNa adds little to clinical decision-making.
🚫 Even for This Narrow Question, FeNa Is Confounded By:
Renders FeNa uninterpretable — increases urinary Na regardless of volume status
Elevated baseline FENa due to impaired tubular reabsorption — prerenal AKI may have FeNa >1%
FeNa can be low despite intrinsic injury — sepsis-associated AKI has mixed hemodynamic and tubular features
Contrast-associated AKI can present with low FeNa, mimicking prerenal disease
Postrenal AKI can have variable FeNa depending on timing and degree of obstruction
FeUrea: Not the Fix You Were Promised
FeUrea was proposed to overcome the diuretic limitation of FeNa. However, diagnostic performance remains poor — sensitivity and specificity are insufficient for clinical decision-making in most scenarios. FeUrea does not reliably distinguish prerenal from intrinsic AKI in diuretic-treated patients.
🔬 Urine Microscopy vs FeNa — Head-to-Head Comparison
🔬 Urine Microscopy (Fresh Specimen)
- Muddy brown casts = ATN. That is the answer.
- RBC casts = glomerulonephritis
- WBC casts = interstitial nephritis
- Hyaline casts = prerenal
- Not confounded by diuretics, CKD, or sepsis
- Provides etiologic diagnosis, not just a number
- Must be fresh (<2 hours) for accuracy
MORE diagnostic information
🧮 FeNa Calculation
- Only answers: prerenal vs ATN?
- Uninterpretable with diuretics
- Misleading in CKD baseline
- False low in sepsis, contrast, early obstruction
- Binary output (high vs low) with no etiologic nuance
- FeUrea does not meaningfully improve performance
- You don't need a calculation when casts give the diagnosis
Commonly over-relied-upon
💡 Bottom Line: Urine microscopy with a fresh specimen gives you MORE diagnostic information than FeNa in virtually every clinical scenario. Muddy brown granular casts = ATN. That is the answer. You do not need a calculation. Teach yourself to spin a urine — it is the single most underused diagnostic tool in nephrology.
🧬 AKI Biomarkers — Now Part of the KDIGO 2026 Definition
KDIGO 2026 formally incorporates structural biomarkers into the AKI definition and staging system (B0/B1 axis). These are no longer just research tools — they are guideline-recommended for clinical use.
🔬 TIMP-2 × IGFBP7 (NephroCheck)
FDA-Approved- Cell cycle arrest markers indicating tubular stress
- Best within 12h of ICU admission or after major surgery/sepsis
- Identifies risk of moderate-to-severe AKI
- Rec 2.4.1 (2B adults, 2C children)
🧪 Urine NGAL
Early Detection- Identifies high risk of severe AKI in hospital settings
- Upregulated within hours of tubular injury (24-48h before SCr rise)
- Useful in cirrhosis-associated AKI to differentiate ATI vs HRS when clinical features are equivocal
📊 Baseline Albuminuria (UACR)
Risk Stratification- Offers additional prognostic value beyond eGFR for evaluating AKI risk
- Useful across inpatient and outpatient settings
- Should be checked in all at-risk patients
⚠️ Important: Urine Eosinophils Are NOT Reliable
Urine eosinophils have LOW sensitivity and moderate specificity — they are limited for EXCLUDING acute interstitial nephritis (AIN). Urinary inflammatory cytokines may be more useful for distinguishing AIN.
🚨 ANURIA: Nephrological Emergency
Definition: Urine output <100 mL per 24 hours
🫀 Vascular Causes
🚰 Obstructive Causes
💊 Furosemide Stress Test (FST)
Revolutionary risk stratification leveraging tubular secretion as a functional marker of tubular integrity.
📋 Protocol
| Dose (naive) | 1.0 mg/kg IV push |
| Dose (prior exposure) | 1.5 mg/kg IV push |
| Timing | Measure UOP over 2 hours |
| Responders | UOP ≥200 mL/2h → lower risk of progression |
| Non-responders | UOP <200 mL/2h → high risk of Stage 3 / RRT |
🎯 Performance
🔥 Intrinsic Renal Disease: Comprehensive Overview
⚡ Rapidly Progressive Glomerulonephritis (RPGN)
NEPHROLOGIC EMERGENCY: Rapid kidney function deterioration with extensive crescent formation
📈 Rising Creatinine
🔬 Active Sediment
⏰ Clinical Urgency
🧬 Categories (by IF)
| Anti-GBM | Linear IgG staining |
| Immune Complex | Granular (lupus, post-infectious) |
| Pauci-Immune | ANCA-associated vasculitis |
🧪 Essential Labs
🔥 Acute Interstitial Nephritis (AIN)
Immune-mediated inflammation of the tubulointerstitium with varied clinical presentations
💊 Common Drug Causes
#1 cause
Omeprazole, pantoprazole
Prescription & OTC
Ibuprofen, naproxen
β-lactams, sulfonamides
Quinolones, rifampin
Thiazides
Loop diuretics
PD-1/PD-L1 inhibitors
Rising incidence
🔬 Diagnostic Features
| Urine | WBC casts, sterile pyuria |
| Timeline | Days to weeks post-exposure |
| FENa | >1% (tubular dysfunction) |
| ⚠️ Urine Eos | NOT diagnostic (poor Sn/Sp) |
💡 Management
Prednisone 1 mg/kg/day × 2-4 weeks
🧬 Acute Tubular Necrosis: Intrinsic Toxins
Endogenous nephrotoxins causing direct tubular injury through various mechanisms
💪 Rhabdomyolysis
- Mechanism: Myoglobin direct toxicity + tubular obstruction
- Triggers: Trauma, drugs, prolonged immobilization, exercise
- Labs: CK >1000 U/L (often >5000), myoglobinuria
- Treatment: Aggressive fluid resuscitation, alkalinization
- Goal UOP: 200-300 mL/hr initially
- Key Point: CK for detection, myoglobin causes actual injury
🩸 Hemolysis
- Mechanism: Free hemoglobin tubular toxicity
- Causes: Transfusion reactions, mechanical hemolysis
- Labs: ↓Haptoglobin, ↑LDH, hemoglobinuria
- Clinical: Dark red/brown urine
- Management: Treat underlying cause, maintain UOP
🧪 Light Chain Nephropathy
- Setting: Multiple myeloma, plasma cell disorders
- Mechanism: Light chain precipitation in tubules
- Microscopy: Large, fractured casts with angular edges
- Diagnosis: Serum/urine immunofixation, free light chains
- Treatment: Chemotherapy, plasmapheresis in severe cases
⚡ Rhabdomyolysis Emergency Protocol
- Normal saline 1-2 L/hr initially
- Goal UOP: 200-300 mL/hr
- Monitor for volume overload
- Sodium bicarbonate in selected cases
- Goal urine pH >6.5
- Avoid if volume overloaded
💊 Antibiotic-Associated Kidney Injury: Comprehensive Guide
Antibiotics represent one of the most common causes of drug-induced kidney injury in clinical practice
| Antibiotic Class | Primary Mechanism | Typical Onset (Days) | Pattern of Injury | Incidence Rate |
|---|---|---|---|---|
| Aminoglycosides | Direct tubular toxicity | 7-10 | Acute tubular necrosis | 10-25% |
| Glycopeptides (Vancomycin) | Oxidative stress, inflammasome activation | 5-10 | Acute tubular necrosis | 5-35% |
| Beta-Lactams | Hypersensitivity reaction | 10-14 | Acute interstitial nephritis | 1-3% |
| Polymyxins | Membrane damage | 5-7 | Acute tubular necrosis | 20-60% |
| Fluoroquinolones | Hypersensitivity reaction | 7-14 | Acute interstitial nephritis | <1% |
| Sulfonamides (Crystalluria) | Crystal formation | 1-3 | Crystal nephropathy | 1-5% |
| Tetracyclines | Direct tubular toxicity | 3-7 | Fanconi syndrome | <1% (modern agents) |
| Macrolides | Hypersensitivity, drug interactions | 7-14 | Acute interstitial nephritis | <1% |
| Amphotericin B | Membrane damage | 5-7 | Acute tubular necrosis | 30-80% |
🧬 Aminoglycosides: Structure-Toxicity Relationship
Key Concept: Nephrotoxicity directly correlates with positive charge and number of amino groups
| Aminoglycoside | Relative Nephrotoxicity | Number of Amino Groups | Positive Charges | Clinical Notes |
|---|---|---|---|---|
| Neomycin | Highest (5/5) | 6 | +6 | Topical use only due to toxicity |
| Gentamicin | High (4/5) | 5 | +5 | Most commonly used, high efficacy |
| Tobramycin | Moderate to High (3/5) | 5 | +5 | Preferred for Pseudomonas |
| Kanamycin | Moderate (3/5) | 4 | +4 | Limited use due to resistance |
| Amikacin | Moderate (2/5) | 4 | +4 | Reserved for resistant organisms |
| Netilmicin | Low to Moderate (2/5) | 3 | +3 | Less nephrotoxic alternative |
| Streptomycin | Lowest (1/5) | 2 | +2 | Primarily ototoxic, less nephrotoxic |
🔬 Mechanism of Charge-Related Toxicity
Higher positive charge → stronger binding to negatively charged phospholipids in proximal tubular cells
More charges → greater megalin-mediated endocytosis → higher intracellular accumulation
Highly charged molecules accumulate more in lysosomes → greater disruption of cellular function
Greater positive charge → stronger binding to mitochondrial ribosomes → more energy disruption
⚖️ Vancomycin vs Aminoglycosides: Comparative Nephrotoxicity
| Aspect | Vancomycin | Aminoglycosides | Clinical Notes |
|---|---|---|---|
| Overall AKI incidence | 5-35% | 10-25% | Varies by definition and population |
| Severe AKI requiring RRT | 1-5% | 2-7% | Higher with prolonged therapy |
| Time to AKI onset | 5-10 days | 7-14 days | Vancomycin often earlier |
| Persistent kidney dysfunction | 5-15% | 10-20% | Higher with advanced age |
| Concomitant use (both drugs) | 35-45% | Synergistic toxicity | |
| Primary mechanism | NLRP3 inflammasome, oxidative stress | Lysosomal disruption, mitochondrial damage | Different subcellular targets |
| Prevention strategy | AUC-guided dosing | Extended-interval dosing | Both reduce toxicity significantly |
🎯 Key Clinical Decision Points
- Vancomycin + Piperacillin-Tazobactam: 35-45% AKI risk (NNH = 8-10 patients)
- Vancomycin + Aminoglycosides: 25-40% AKI risk (avoid when possible)
- AUC-guided vancomycin dosing: 33-45% reduction in AKI risk
- Extended-interval aminoglycosides: 30-50% reduction in nephrotoxicity
⚠️ High-Risk Antibiotic Combinations
Synergistic nephrotoxicity from commonly used antibiotic combinations
🔥 Vancomycin + Piperacillin-Tazobactam
- AKI Risk: 21-40% (vs 8-13% vancomycin alone)
- Mechanism: Synergistic NLRP3 inflammasome activation
- Risk Factors: Age >65, CKD, diabetes, high doses
- Prevention: AUC-guided vancomycin + extended-infusion pip-tazo
- Alternatives: Vancomycin + cefepime or meropenem
⚡ Vancomycin + Aminoglycosides
- AKI Risk: 25-40% (historic high-risk combination)
- Mechanism: Complementary nephrotoxic pathways
- Risk Factors: Higher doses, extended duration, pre-existing CKD
- Management: Avoid combination when possible
- Monitoring: Daily creatinine, enhanced biomarker surveillance
💀 Polymyxins + Vancomycin
- AKI Risk: 40-60% (extremely high-risk combination)
- Mechanism: Synergistic membrane damage + oxidative stress
- Indication: Extensively drug-resistant organisms only
- Management: Nephroprotective strategies, daily monitoring
- Alternatives: Consider newer agents when available
🚨 Triple Combination Therapy
- AKI Risk: 45-70% (vancomycin + aminoglycoside + beta-lactam)
- Mechanism: Multiple complementary nephrotoxic pathways
- Risk Factors: Nearly universal in high-risk patients
- Management: Avoid when possible, daily monitoring, early de-escalation
- Rule: Each additional nephrotoxin increases AKI odds by ~60%
🏭 Environmental & Heavy Metal Toxicity
⚗️ Ethylene Glycol
- Source: Antifreeze ingestion
- Mechanism: Toxic metabolites (oxalic acid)
- Timeline: 6-12 hours post-ingestion
- Treatment: Fomepizole, hemodialysis
- Lab findings: Anion gap metabolic acidosis
🔶 Heavy Metals
- Mercury: Proximal tubular necrosis
- Lead: Chronic tubulointerstitial disease
- Cadmium: Proximal tubular dysfunction
- Treatment: Chelation therapy (DMSA, EDTA)
- Monitoring: 24-hour urine metals
🍄 Natural Toxins
- Mushroom poisoning: Amanita species
- Snake venom: Hemolysis, direct nephrotoxicity
- Aristolochic acid: Chinese herbs (chronic)
- Treatment: Supportive care, specific antidotes
- Prevention: Education, avoid herbal remedies
💎 Crystalopathy: Crystal-Induced Tubular Obstruction & AKI
Pathophysiology: Intratubular crystal precipitation causing mechanical obstruction, direct tubular toxicity, and acute kidney injury
🔬 Common Crystalopathy Mechanisms
- Exceed solubility limits in tubular fluid
- Concentration-dependent precipitation
- pH-dependent solubility (uric acid, cystine)
- Temperature effects on crystal formation
- Mechanical blockage of tubular lumens
- Increased intratubular pressure
- Reduced effective filtration
- Secondary tubular cell injury
- Crystal-induced inflammation
- Complement activation
- Reactive oxygen species generation
- Tubular epithelial cell death
🚨 Tumor Lysis Syndrome (TLS): Life-Threatening Uric Acid Crystalopathy
Most dangerous crystalopathy - requires immediate recognition and intervention within hours
⚡ Critical Recognition Triad
- Burkitt lymphoma (highest risk)
- High-grade NHL with bulky disease
- ALL/AML with high WBC count
- Recent chemotherapy initiation
- Hyperuricemia (>8 mg/dL)
- Hyperkalemia (>6.0 mEq/L)
- Hyperphosphatemia (>4.5 mg/dL)
- Hypocalcemia (<7.0 mg/dL)
- Peak risk: 12-72 hours post-chemo
- Hyperkalemia most immediately lethal
- Can progress to AKI requiring RRT
- Prevention superior to treatment
💊 Emergency Management Priorities
- Cardiac monitoring for hyperkalemia
- STAT electrolytes, uric acid, phosphorus
- G6PD testing if rasburicase planned
- Assess volume status
- Hyperkalemia treatment if K+ >6.5 mEq/L
- Rasburicase 0.2 mg/kg IV if indicated
- Aggressive hydration if not volume overloaded
- Nephrology consultation
- Serial electrolytes Q6-8h
- Monitor for AKI development
- Prepare for RRT if refractory
- Avoid calcium if PO4 >6.5 mg/dL
🎯 TLS Key Clinical Pearls
- Burkitt lymphoma: Highest risk malignancy
- High-grade NHL: Risk increases with tumor burden
- ALL/AML: WBC >50,000 or bulky disease
- Timing: Peak risk 12-72 hours post-chemo
- Mechanism: Converts uric acid → allantoin (water-soluble)
- Contraindications: G6PD deficiency (hemolysis risk)
- Efficacy: Uric acid normalizes within 4 hours
- Monitoring: Uric acid levels q6h
- Calcium administration: Risk of Ca-PO4 precipitation
- Inadequate hydration: Must achieve high UOP
- Delayed recognition: Monitor high-risk patients proactively
- Urine alkalinization: Not recommended (may worsen Ca-PO4)
- Baseline: BMP, uric acid, phosphorus, LDH
- Frequency: Q6-8h x 72 hours minimum
- AKI monitoring: Daily creatinine, urine output
- Response: Uric acid should decline within 24h
⚠️ TLS Prevention Strategies
- Tumor burden assessment (CT, PET scan)
- Baseline electrolytes, uric acid, LDH
- G6PD testing if rasburicase anticipated
- Renal function and volume status
- Allopurinol 300mg daily x 1-2 days pre-chemo
- Aggressive hydration (3-4 L/day if tolerated)
- Rasburicase prophylaxis in very high-risk patients
- Consider modified chemotherapy regimen
- Electrolytes Q6h x 72 hours minimum
- Daily weights and strict I/O monitoring
- Continuous cardiac monitoring if K+ >6.0
- Early nephrology involvement for high-risk cases
💎 Other Important Crystallopathies in AKI
Additional crystal-induced kidney injury patterns requiring specific recognition and management
🎯 Uric Acid Crystalopathy (Non-TLS)
- Causes: Gout flares, dehydration, acidic urine (pH <5.5)
- Mechanism: Uric acid precipitation in acidic tubular fluid
- Recognition: Yellow-brown needle-shaped crystals
- Treatment: Alkalinization (target pH 6.5-7.0), hydration
- Prevention: Allopurinol for chronic hyperuricemia
🧡 Calcium Oxalate Crystalopathy
- Causes: Ethylene glycol poisoning, high-dose vitamin C
- Mechanism: Oxalate overproduction or ingestion
- Recognition: Envelope-shaped crystals, anion gap acidosis
- Treatment: Fomepizole (ethylene glycol), hemodialysis
- Timeline: AKI develops 6-12 hours post-ingestion
💊 Drug-Induced Crystallopathy
- Acyclovir: Rapid IV infusion, dehydration
- Sulfonamides: Crystalluria in acidic urine
- Methotrexate: High-dose therapy, delayed excretion
- Indinavir: HIV protease inhibitor crystallopathy
- Prevention: Adequate hydration, appropriate infusion rates
⚡ Calcium Phosphate Precipitation
- Triggers: Rapid calcium administration + high phosphate
- Risk factors: CKD, phosphate retention, alkalotic urine
- Recognition: Sudden AKI after calcium/phosphate administration
- Prevention: Avoid calcium if phosphate >6.5 mg/dL
- Clinical context: Common complication in TLS management
🔴 Cystine Crystalopathy
- Cause: Cystinuria (genetic defect in amino acid transport)
- Recognition: Hexagonal crystals, family history
- Management: Alkalinization (pH >7.0), high fluid intake
- Medications: Tiopronin, penicillamine for stone prevention
- Complication: Recurrent nephrolithiasis and AKI
🔬 2,8-Dihydroxyadenine Crystalopathy
- Cause: Adenine phosphoribosyltransferase (APRT) deficiency
- Recognition: Mimics uric acid stones, genetic testing
- Treatment: Allopurinol (blocks adenine metabolism)
- Importance: Often misdiagnosed as uric acid crystalopathy
- Outcome: Excellent response to allopurinol if recognized
🎯 General Crystallopathy Management Principles
- Target UOP 2-3 mL/kg/hr when possible
- Monitor for volume overload in CKD patients
- Consider loop diuretics if fluid retention
- Maintain euvolemia while maximizing clearance
- Uric acid: Alkalinize urine pH >6.5
- Cystine: Alkalinize urine pH >7.0
- Calcium phosphate: Avoid alkalinization
- Monitor: Urine pH q6h during treatment
- Identify high-risk patients early
- Prophylactic hydration for procedures
- Appropriate drug dosing and infusion rates
- Monitor for drug interactions affecting clearance
- Serial creatinine and urine output
- Urine microscopy for crystal identification
- Electrolyte panel q6-8h in acute phase
- Specific markers (uric acid, oxalate) as indicated
🏥 Contrast-Associated AKI (CA-AKI)
Evolution from "Contrast-Induced": Recognition of multifactorial pathogenesis beyond direct contrast toxicity
⚡ Risk Factors
- Primary: CKD (eGFR <60), diabetes, volume depletion
- Procedural: High contrast volume, intra-arterial route
- Patient: Age >75, heart failure, multiple myeloma
- Concurrent: Nephrotoxic medications, hypotension
🛡️ Prevention Strategies
- Hydration: Isotonic saline 1-1.5 mL/kg/hr × 6-12h
- Contrast: Minimize volume, use iso/low-osmolar agents
- Medications: Hold nephrotoxins, avoid NSAIDs
- Timing: Space procedures ≥48-72 hours apart
📊 Management Pearls
- Timeline: AKI develops 24-72h post-exposure
- Peak: Creatinine peaks at 3-5 days
- Recovery: Usually complete within 1-2 weeks
- Monitoring: Serial creatinine, urine output
🧮 AKI Assessment Tools
Enhanced Drug Nephrotoxicity Risk
KDIGO Staging Calculator
Furosemide Stress Test
CA-AKI Risk Assessment
🗺️ AKI Diagnostic Flowchart
↑Creatinine (≥0.3 mg/dL or 1.5× baseline) OR ↓UOP (<0.5 mL/kg/hr × 6h)
KDIGO Staging + Check for life-threatening complications (hyperkalemia, acidosis, volume overload)
History + Exam + Urine microscopy + Laboratory assessment + Imaging if indicated
Address underlying cause + Supportive care + Monitor for recovery vs progression
Stage 2-3 AKI + Unclear etiology + Need for RRT + Complicated cases
🎯 Essential AKI Learning Points
🚨 Emergency Recognition
- Anuria = nephrological emergency
- Check hyperkalemia immediately
- Foley catheter if obstruction suspected
- Fresh urine microscopy <2 hours
🔬 Diagnostic Essentials
- Urine microscopy differentiates causes
- FENa <1% suggests prerenal
- Muddy brown casts = ATN
- RBC casts = glomerulonephritis
🧪 Advanced Tools
- FST predicts progression/RRT need
- Superior to biomarkers
- KDIGO staging guides management
- CA-AKI is multifactorial
💡 Clinical Pearls
- AKI-AKD-CKD continuum concept
- Prevention better than treatment
- Early nephrology consultation
- Monitor for recovery patterns
🔥 Intrinsic Disease
- RPGN = nephrologic emergency
- Classic AIN triad rarely present
- Urine eosinophils NOT diagnostic
- Drug withdrawal first-line for AIN
💊 Drug Nephrotoxicity
- Aminoglycosides: delayed onset (5-10 days)
- Vancomycin: trough level dependent
- PPIs most common cause of AIN
- Multiple nephrotoxins = exponential risk
🛡️ Multicomponent Kidney-Protection Strategy (KDIGO 2026)
Figure 9 — Escalating Prevention Bundle: For patients at high risk through AKI Stage 3
Review NSAIDs, aminoglycosides, contrast, ACEi/ARB in acute setting
Targeted resuscitation, avoid both hypo- and hypervolemia
Especially in critically ill or hemodynamically unstable patients
Serial assessment to detect progression early
Renally cleared medications require dose adjustment as GFR changes
Urine microscopy, imaging, serologies based on clinical context
Refractory hyperkalemia, acidosis, volume overload, uremic symptoms
💧 Fluid Management in AKI (KDIGO 2026)
Updated evidence-based recommendations for volume resuscitation and hemodynamic targets
💉 Volume Resuscitation
Crystalloids Over Colloids
Use crystalloids over colloids (albumin, gelatin, starches) for volume expansion in AKI risk or active AKI.
Buffered Crystalloids Preferred
Buffered crystalloids (LR, Plasmalyte) over 0.9% NaCl except in traumatic brain injury. This is a strong, new preference in KDIGO 2026.
Severe Metabolic Acidosis
pH <7.20 → IV bicarbonate over RRT unless other urgent RRT indication exists.
Liberal IV Fluids for Major Surgery
Liberal IV fluids (positive balance 1-2 kg at 24h) over restrictive (net zero) for elective major abdominal surgery.
🫀 Hemodynamic Targets
MAP Target >65 mmHg
Target MAP >65 mmHg in critically ill patients. Individualize: higher for chronic HTN, lower for chronic hypotension.
No Low-Dose Dopamine
Recommend AGAINST low-dose dopamine for kidney protection. Grade 1A — strongest possible recommendation.
⚖️ Protein in AKI — Avoid Both Extremes
Neither protein restriction nor high-protein loading is appropriate. The evidence supports a balanced approach.
Protein Restriction
- Worsens malnutrition and catabolism
- Does NOT prevent or delay RRT
- Does NOT preserve kidney function in AKI
- Increases muscle wasting in critical illness
THE
SWEET
SPOT
Adequate protein to prevent catabolism without excessive load on injured kidneys
High-Protein Loading
- No survival benefit (large pragmatic RCT)
- Higher mortality in AKI patients
- Longer time to hospital discharge
- Increases urea generation and uremic symptoms
📈 The High-Protein Harm Signal
A large pragmatic RCT compared high protein (≥2.2 g/kg/day) vs standard (≤1.2 g/kg/day). AKI patients randomized to high protein had:
- Higher mortality
- Longer time to discharge
- Increased urea generation → worsened uremic symptoms
- May accelerate the need for RRT
🎯 Practical Guidance
- Goal: Adequate protein to prevent catabolism — not more
- Caloric targets do NOT change for AKI unless on RRT (PP 3.9.4)
- On RRT: Protein losses through dialysis circuit may require modest increase
- Involve nutrition team early for critically ill AKI patients
- Monitor: Prealbumin trends, nitrogen balance if available
💡 Bottom Line: Protein restriction does not protect injured kidneys. High-protein loading harms AKI patients. Feed normally — provide adequate calories and moderate protein to prevent catabolism without generating excessive urea load. This is one of those areas where “more is better” thinking gets patients in trouble.
📋 Post-AKI Follow-Up Care (KDIGO 2026 Chapter 6 — NEW)
Entirely new chapter in KDIGO 2026 — structured approach to AKI survivorship
🤝 Coordinated Follow-Up
- All AKI/AKD survivors need coordinated follow-up with personalized care
- Clear transition of care with identified healthcare professional
- AKI episode details
- Resolution status
- Medication management
- Follow-up plan
🩺 Post-AKI Comprehensive Assessment
💊 Post-AKI Medications
- RASi (ACEi/ARB) unless contraindicated
- Evaluate for SGLT2i
- Evaluate for GLP-1 RA
- Evaluate for MRA
- Assess at discharge
- Reassess by 3 months
- Monitor per KDIGO 2024 CKD guideline
🧬 CKD Evaluation
- Assess kidney function and damage markers 3 months after AKI/AKD
- Use cystatin C-based GFR when SCr is less accurate (muscle wasting, critical illness recovery)
🏥 WATCH-ME Framework — Patients Discharged on Dialysis
🤒 Sick Day Protocols
Temporarily stop specific drugs during acute illness with volume depletion risk
KDIGO 2026 Alert Guidance (Rec 2.2.1, 1B)
Interruptive creatinine-based electronic alerts for AKI in isolation, WITHOUT systematic strategies for response, are recommended AGAINST. Alerts must trigger a structured response bundle (see Prevention Strategy above), not just a notification. An alert without a protocol is noise — an alert with a bundle saves kidneys.